Our Facility offers state-of-the-art technologies that enable visualization, digitization, and image analysis. We provide comprehensive one-stop imaging services that include technology- implementation instructions for image acquisition and analysis. The team of scientists and trained technicians works in close collaboration to advance our science seamlessly across fields and technologies.
Tissue samplimg, staining and labeling

The histology laboratory offers expertise in processing and staining tissues. Tissue slides are prepared for analysis and for confirming the diagnosis of pathologic conditions to support investigators in their studies. The facility offers standard H&E or other, specific tissue staining upon request. Extensively trained technicians can provide paraffin-embedded sections or frozen sections for the best histochemistry and immunohistochemistry results. We process fixed tissues by embedding them in paraffin or OCT compound as blocks. We section the tissues and place each section on a slide directly or through the use of the floating section method. Customization is done upon request after consultation with the investigator to set up the optimal conditions for a successful result.
Light Microscopy Center
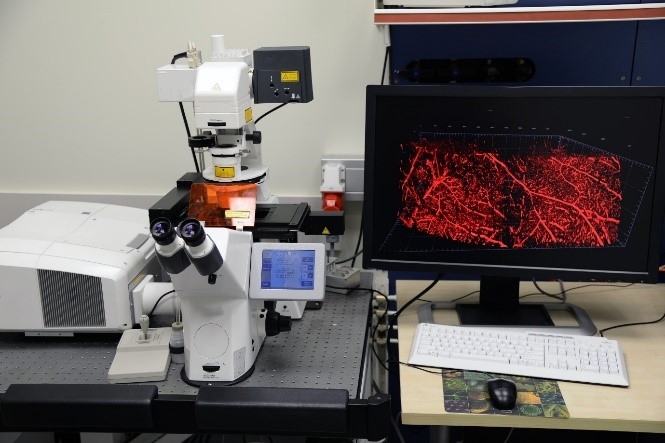
Our light microscopy center houses laser scanning confocal (Confocal LSM 710) and Stereomicroscopes. We offer Imaging-based approaches using state-of-the-art techniques (such as Clearing methods) to localize proteins at the subcellular level. This is done either through the use of specific primary antibodies and fluorophore-carrying secondary antibodies, or with proteins attached to a fluorescent protein, thus enabling temporal studies of the protein’s distribution in its natural environment.
Image analysis
We provide and support a wide variety of image analysis and visualization tools. We assist in establishing data-analysis protocols. We use FIJI/ImageJ (NIH) for Image analysis and process, Zen 2010 (Zeiss) Offline full version for 3D reconstruction, 2D colocalization and processing, Image-Pro Premier (Media Cybernetics) for segmentation, classification, count, measurements for brightfield and 2D fluorescence images and Adobe Photoshop (Adobe) for Image, line art and graph juxtaposition for visualization and preparation for publication.
H2-Histology and Pathology Gallery

A. Nissl stain of rat brain.
B. PAS staining for conjuctival goblet cells (Goblet cells, Purple).
C. Confocal image of cilia in rat trachea (β-tubulin, Green).
D. Confocal image of rat lung / alveolar epithelium (T1α, TypeI, Green; SPD, TypeII, Red).
E. Transparent whole mouse brain following clearing procedure.
F. Confocal image of neurons (Thy1 transgenic mice) following clearing procedure of whole mouse brain.
G. Confocal image of blood vessels (Vecadcre mice (B6.FVB-Tg(Cdh5-cre)7Mlia/J, Jackson) crossed with tdTomato flox/stop/flox mice (B6.Cg-Gt(ROSA)26Sor tm14(CAG-tdTomato)Hze/J , Jackson) following clearing procedure of whole mouse brain.
H. Confocal image of hipocampal axons and dendrites (MAP2, Red; Nuclei, DAPI) of rat brain.
I. Confocal image of astrocytes (GFAP, Red) and viable neurons (NeuN, Green) of rat brain.
J. Confocal image of a single neuron in the Piriform Cortex area of rat brain following clearing procedure.
K. Confocal image of resting astrocytes in rat brain (GFAP, Red.)
L. High magnification of hipocampal axons and dendrites (MAP2, Red; Nuclei, DAPI) in the rat brain.

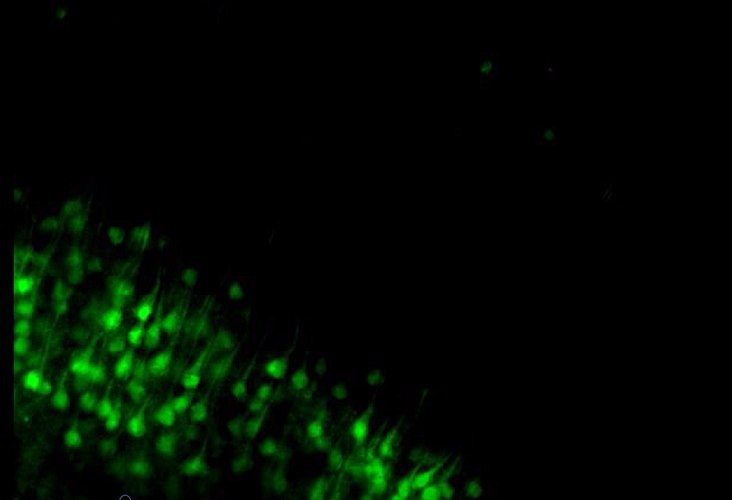
Movie: Mouse brain cortex from the 1 eYFP line following clarity process

 Movie: Immunostaining_of_rat_alveolar_type_I_and_II_cells_(3D)
Movie: Immunostaining_of_rat_alveolar_type_I_and_II_cells_(3D)