Pre-clinical animal research is key in experimental biology and biomedicine. The design and selection of animal models are form the basis for the validity of scientific experiments, as well as for the careful extrapolation of results to human biology. Researchers at the IIBR are developing specialized animal models for diagnostics, vaccine development and therapy. The work is carried out according to Israeli guidelines for animal welfare, while searching for new ways to replace animal experiments with alternative methods, such as cell culture, when possible. Some of our current projects include models for ricin, Botulinum toxin intoxication and Vaccine development
Ricin-induced ARDS in Swine:
Scientists at the IIBR established a novel animal model for the study of ricin-induced acute respiratory distress syndrome (ARDS). This model, based on intratracheal instillation of ricin to swine, is compatible with barometric whole-body plethysmographic surveillance of non-anesthetized animals, profiling PaO2/FiO2 ratios over time in anesthetized mechanically-ventilated animals and evaluating IC measures.
Innovative animal model for the study of Butulinum Toxin:
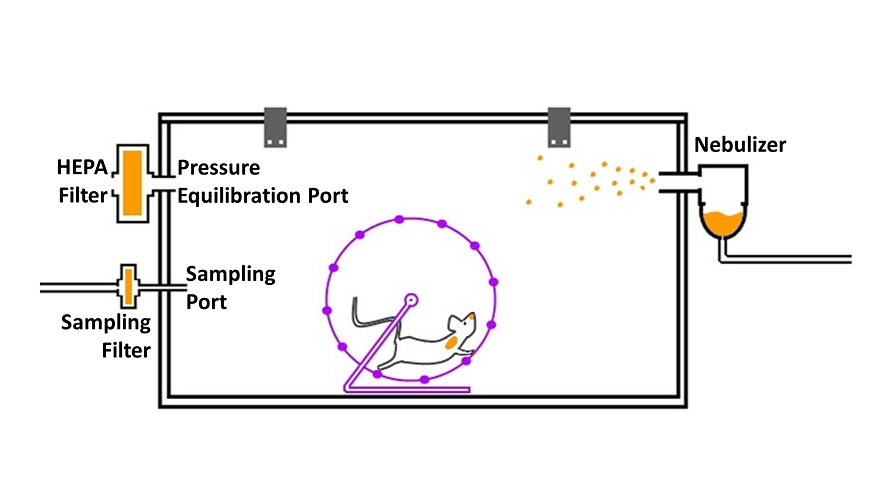
Clinical data suggest that the therapeutic window for many infectious diseases is limited. Evaluating the efficacy of current and innovative therapeutics after the manifestation of clinical symptoms is therefore of great interest. Scientists at the IIBR are developing advanced animal models to study real-time therapeutic efficacy. These novel experimental systems combine hardware and in-house written software for real time physiological analysis that enable detection of specific physiological biomarkers long before any conventional visual symptoms can be detected.


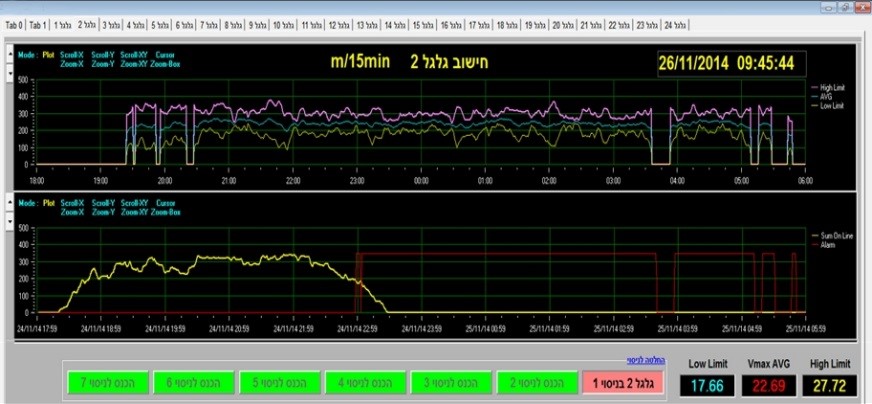
Figure 1: Scientists at the IIBR develop novel physiological monitoring methods to improve the detection of early poisoning symptoms. TOP: Rabbit normal respiration profile. Deviation from this profile were validated as early symptoms of botulism that were used as a successful ‘trigger to treat BOTTOM: By using advanced signal analysis, IIBR scientists, detect very early effects, in mouse treadmill-running patterns. This analysis is the basis of IIBR’s sensitive animal model for botulism and for the efficacy of related therapeutics.
Modeling Eczema vaccinatum in atopic dermatitis mice
Eczema vaccinatum is a severe adverse reaction of smallpox vaccine highly associated with a background of atopic dermatitis. Following 9 weeks of dinitrofluorobenzene (DNFB) treatment, either SKH-1 hairless mice or Nc/Nga mice develop severe symptoms of atopic dermatitis. Poxvirus (ectromelia virus) dermal exposure results in severe viral disseminated highly resembling human eczema vaccinatum. Effective treatment was obtained by combined antibodies and antiviral treatment. Active vaccination with Modified Vaccinia Ankara virus (MVA) induced protective immunity despite aberrant dermatitis immune response.
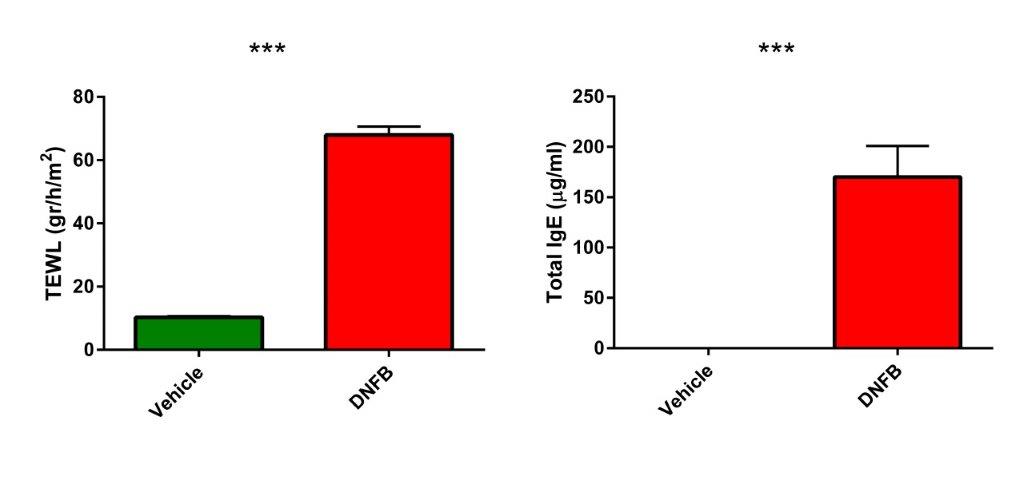
Measures of atopic dermatitis in DNFB treated SKH-1 mice:
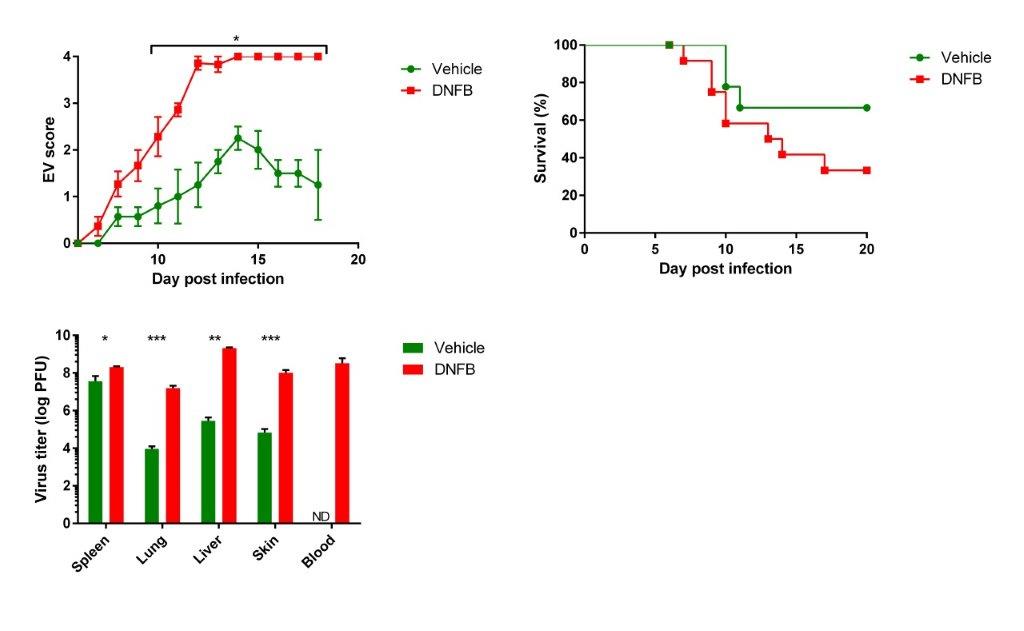
Measures of Eczema vaccinatum (Eczema score, survival and viral load) in ectromelia virus infected atopic dermatitis SKH-1 mice.
Efficacy of Inhalational Gentamicin Treatment in a Pneumonic Plague Murine model
Pneumonic plague is characterized by a rapid and fulminant development of acute pneumonia and septicemia resulting in death within days of exposure and infection. Antibiotic administration is the main treatment, effective if given within a narrow therapeutic window spanning from exposure till ~24hrs after symptom onset. These symptoms are non-specific and "flu-like" in nature, hindering correct diagnosis and treatment. This could result in delayed diagnosis leading to an unavoidable loss of life for the initial patients'. These facts motivate research into improving treatment options for patients with significant disease burden.
In this research project we are evaluating the efficiency of inhalational gentamicin therapy against pneumonic plague. This administration route could allow the rapid administration of clinically significant drug doses directly to the main site of infection, possibly improving late-stage patients' survival. Our preliminary results indicate that the inhalational treatment with gentamicin was highly effective against pneumonic plague and allowed rapid clearance of the pathogen from the infected lung as demonstrated by whole body imaging analysis using the IVIS system.