Francisella tularensis, the causative agent of tularemia, is a gram-negative facultative intracellular bacterium that can cause a life-threatening infection in humans. Depending on the site of infection, tularemia has several characteristic clinical variants. Of these, the respiratory route of infection is the most severe, causing a deadly disease upon exposure to a very low inhalation dose (as few as 1–10 bacteria). Due to its low infectious dose, ease of spread by aerosol, and high virulence, F. tularensis is classified as a Tier 1 Select Agent by the U.S. government, and as a potential bio-terror agent.
IIBR scientists are interested in understanding the mechanisms of protection against F. tularensis infections through studying the pathogenesis of this bacterium and the host-pathogen interactions. In that contest, we recently investigated the recall immune response, following vaccination with an attenuated strain (LVS). In-vivo analysis of the immune response subsequent lethal challenge, demonstrated that activation of T cells, reflected by the enhanced transcriptional response of cytokines such as IFN-gamma and IL-17, is initiated as early as 24 hours post-challenge by lung T cells. This immediate response restricts the initial dissemination of the invading bacteria from the lungs to internal organs. Thus, the early recall response taking place in the lung extends the time window necessary for later phases of the protective response.
Consequently, future prophylactic strategies to counteract F. tularensis infection, may require modulation of the immune response within the lung.
In another line of research, we described an unbiased bacterial whole-genome immunoinformatic analysis aimed at selection of potential CTL epitopes located in “hotspots” of predicted MHC-I binders. Applying this approach to the proteome of F. tularensis resulted in identification of 170 novel CTL epitopes, several of which were shown to elicit highly robust T cell responses. We further demonstrated that immunization using six of the most prominent identified CTL epitopes, induced a potent and specific CD8+ T cell response. This CTL-specific mediated immune response prevented disease development, allowed for a rapid clearance of the bacterial infection and provided protection against a lethal challenge with F. tularensis. Our approach provides a proof-of-principle for selecting and generating a multi-epitope CD8+ T cell-stimulating vaccine against a model intracellular bacterium.
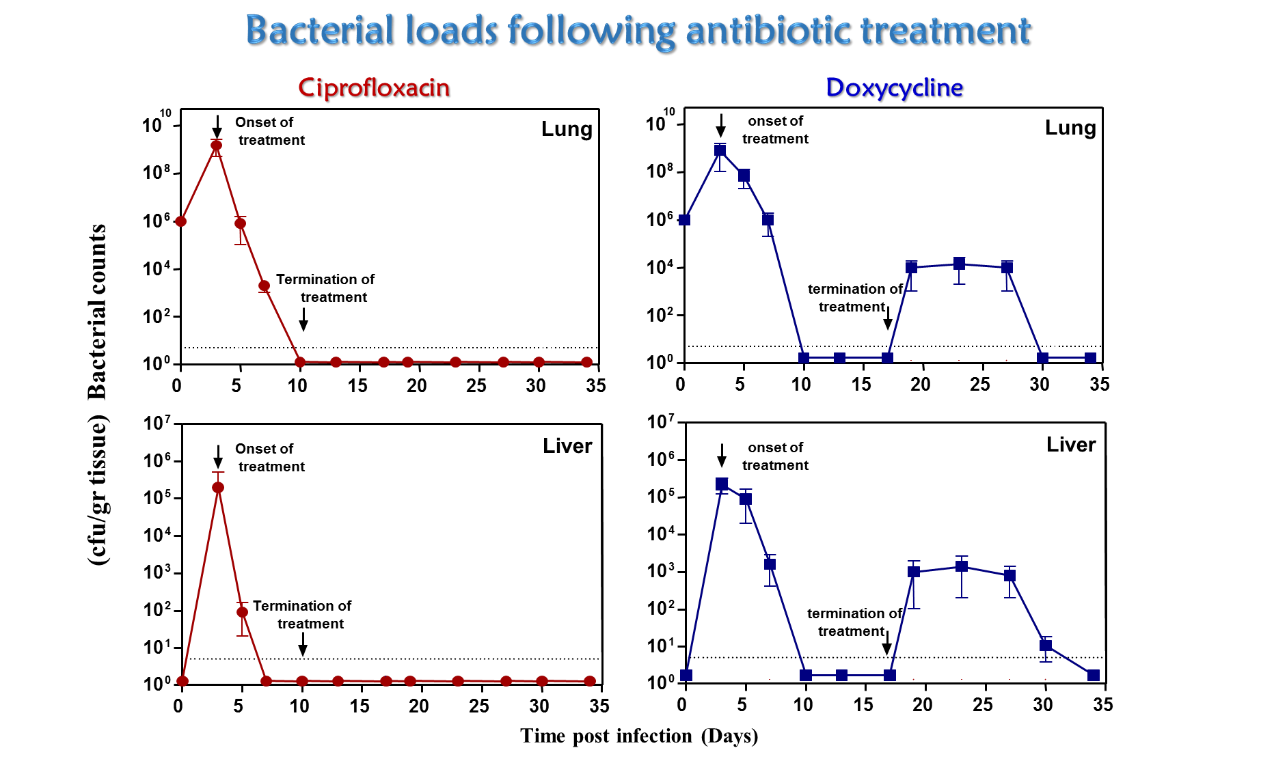
Antibiotics usually provide curative therapy for tularemia. However, mortality depends on the infection type, patient health, antibiotics selected for treatment and treatment onset. We established a murine model system to evaluate the efficacy of antibiotic treatment against F. tularensis. We found that both ciprofloxacin and doxycycline are effective in preventing the development of tularemia in the mouse model following airway infection, but in the case of bacteriostatic antibiotics such as doxycycline there is a significant failure to achieve complete recovery unless treatment is initiated within a short time post-infection. At later stages of the disease, prolonging ciprofloxacin treatment but not prolonging doxycycline treatment appeared to be an effective strategy for successful therapy against the highly virulent F. tularensis strain. These observations have important implications for designing efficient therapeutic approaches for treatment of tularemia in various scenarios where early treatment is not possible.